Functional Materials
Ion Conductive Materials
"Ion conductive materials" holding the key to next-generation energy
The Niterra Group develops "ion conductive materials" in which electricity flows as specific ions move at high speeds within the ceramic crystal lattice.
Reasons why ion conductive materials are chosen-
1.Oxygen ion conductors (oxide-based)
Oxygen ions move within the crystal lattice. Leveraging technologies cultivated over many years, we contribute to improving the performance of oxygen sensors and SOFCs (Solid Oxide Fuel Cells), which are next-generation power generation devices. -
2.Lithium ion conductors
Lithium ions move within the crystal lattice. By using them as "solid electrolytes" in applications such as all-solid-state batteries, we solve challenges including extending battery life, ensuring safety, and improving energy density.
Examples of use within the Niterra Group
Solid electrolyte materials for oxygen sensors
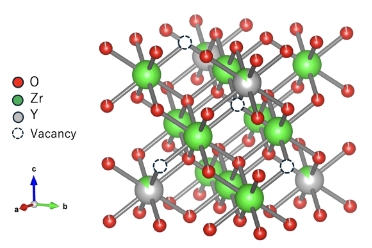
Achieving both high conductivity and low thermal expansion through valence control
This material achieves high oxygen ion conductivity by generating oxygen ion (O²⁻) vacancies in the material through a valence control technology that replaces Zr⁴⁺ in zirconia (ZrO₂) with Y³⁺. Particularly in oxygen sensors that require a bonded structure with alumina components, it also features a low thermal expansion coefficient comparable to that of alumina.
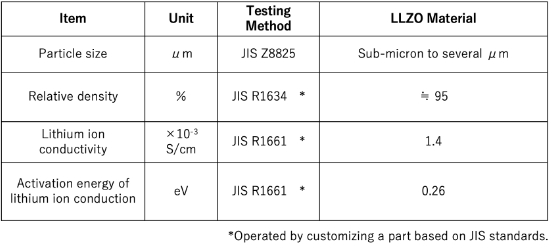
Solid electrolyte materials for Li-ion batteries
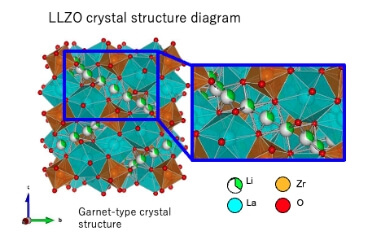
"LLZO," a next-generation lithium ion conductive material that achieves the highest level of conductivity among oxide-based materials
The Niterra Group's "LLZO (Lithium Lanthanum Zirconate)" is a solid electrolyte material boasting world-class lithium ion conductivity among oxide-based materials. We created "gaps" that serve as ion pathways by partially replacing lithium (Li) in the crystal lattice with magnesium (Mg), and further expanded the crystal lattice spacing by replacing lanthanum (La) with strontium (Sr).
This unique crystal structure control enables the high-speed movement of lithium ions. Provided in an easy-to-handle powder form, it can be utilized for next-generation all-solid-state batteries, greatly contributing to longer battery lifespans and improved safety. Through atomic-level design that draws out the potential of materials, we are opening up new possibilities for energy storage devices.
Electron Conductive Materials
"Electron conductive materials" that achieve metal-like electrical conductivity with ceramics
"Electron conductive materials" equipped with metal-like conductivity allow electricity to flow through the movement of electrons and holes within ceramics, which are typically insulators. Generally, in oxides (such as TiO₂ and ZnO), lattice defects and impurities cause electrons and holes to act as carriers, allowing them to function as semiconductors. Certain nitrides (such as TiN) and carbides (such as SiC) also exhibit conductivity.
Reasons why electron conductive materials are chosen
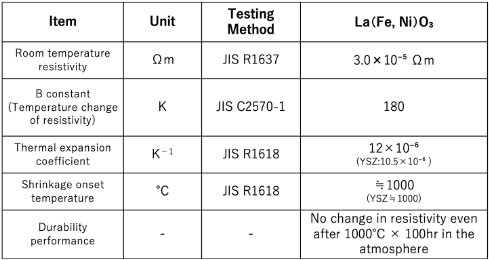
The LaNiFeO₃-based conductive material,
used as an electrode and lead material in zirconia oxygen sensors operating in high-temperature environments, is stable even at high temperatures. It is also characterized by a thermal expansion coefficient close to that of the base zirconia ceramics, which suppresses heat-induced degradation and peeling.
LaNiFeO₃-based conductive materials
Highly durable electrode materials supporting the reliability of oxygen sensorsThe LaNiFeO₃-based conductive material, used as an electrode and lead material in zirconia oxygen sensors operating in high-temperature environments, is stable even at high temperatures. It is also characterized by a thermal expansion coefficient close to that of the base zirconia ceramics, which suppresses heat-induced degradation and peeling.
Piezoelectric/Dielectric Materials
"Dielectric and piezoelectric materials" that control the charge polarization of ceramics
Reasons why dielectrics are chosenDielectrics are a type of "insulator" that do not conduct electricity. They have the property of slight internal charge polarization when an external voltage (electric field) is applied. Due to this characteristic, materials such as barium titanate (BaTiO₃) and strontium titanate (SrTiO₃) are widely used in electronic circuits as capacitors and insulators.
Reasons why piezoelectrics are chosenAmong dielectrics, materials that generate voltage when subjected to external pressure (direct piezoelectric effect) and conversely deform when a voltage is applied (inverse piezoelectric effect) are called piezoelectrics. Representative materials include ceramics such as lead zirconate titanate (PZT). Leveraging their conversion functions, they are utilized in ultrasonic sensors, actuators, ignition devices, and more.
Piezoelectric materials for knock sensors
Piezoelectric materials combining high output performance and high-temperature stabilityThis is a piezoelectric material based on lead zirconate titanate (Pb(Zr,Ti)O₃) that exhibits high output and excellent high-temperature stability. Through proprietary composition control, it achieves both a high output with a piezoelectric constant d₃₃ of 340 pC/N or more and high-temperature stability to withstand harsh environments. By using it in knock sensor elements, it greatly contributes to making the sensor itself lighter and improving its durability.

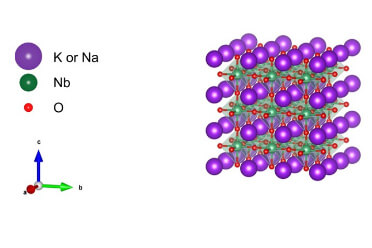
Lead-free piezoelectric materials for actuators/sensors
Lead-free and compliant with the RoHS Directive. Promotes device evolution with a 40% weight reduction compared to PZT (lead zirconate titanate).This is a KNN (alkali niobate)-based lead-free piezoelectric material. Because it contains no lead, it can be easily disposed of even after lead regulations (e.g., the RoHS Directive) are implemented. It is lighter (-40%) than the existing material, PZT (lead zirconate titanate), and is expected to reduce the weight of various components.